|
|
Organic ChemistryUsing Molecular Models |
| Return to Index | Go to next page |
Three resources will help you learn to use your model kit: the kit manual, which has many good color pictures; demonstration by the instructor; and online molecular models, which will allow you to compare the model you build to a more conventional representation. Demonstrations and online models will be provided throughout the course; reading the manual is, of course, up to you!
Some of the laboratory periods will be devoted to learning to use your model kit, and you will be required to identify molecular models during examinations.
Your kit contains several different types of pieces, but the ones which concern us at present are those which allow us to represent organic molecules. These are the tetrahedral pieces, the gray double-bond end-pieces which go with the double bond pieces, and the triple bonds. There are also several colored balls.
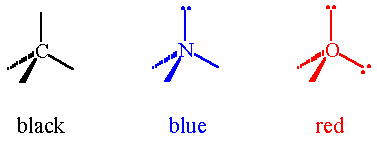  |
Tetrahedral pieces should not be confused with the gray double-bond pieces! There are black, red and blue tetrahedral pieces, which are the standard (or CPK) colors for carbon, oxygen and nitrogen respectively. To assemble a tetrahedral center, take two tetrahedral pieces (usually of the same color) and snap them together. A tetrahedral center represents an atom with four groups attached. Remember from VSEPR that a group can be either a bond or a lone pair of electrons.
Assemble tetrahedral centers from tetrahedral pieces of each color. Notice that the four arms or "bonds" are arranged at angles of 109.5°. The black center represents a carbon atom, with four bonds. The blue center represents a nitrogen atom, with three bonds and a lone pair of electrons. The red center represents an oxygen atom, with two bonds and two lone pairs.
| Return to Index | Go to next page |

|
Dan Berger's home page |
|
Dan Berger's chemistry pages |
|
Bluffton organic chemistry pages |
Copyright © 1998, 1999, 2000, 2003, 2007 by Daniel J. Berger. This work may be copied without limit if its use is to be for non-profit educational purposes. Such copies may be by any method, present or future. The author requests only that this statement accompany all such copies. All rights to publication for profit are retained by the author.