|
|
Organic ChemistryMolecular Models:
|
| Return to previous page | Return to Index | Go to next page |
1. Alkanes, alkenes and alkynes
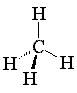
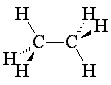
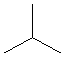
Review the sections in your texts concerning alkane nomenclature. Using your model kit, build methane, ethane, propane and butane and compare them to the online models. Rearrange the model of butane to its constitutional isomer, 2-methylpropane (isobutane). This demonstrates that constitutional isomers use the same atoms, differently connected. In the table below, line drawings have been used for butane and 2-methylpropane. Line drawings cannot be used for methane and are almost never used for ethane or propane. But line drawings make higher alkane structures much easier to read!
 |
 |
 |
||
 |
 |
 |
 |
 |
| Return to previous page | Return to Index | Go to next page |

|
Dan Berger's home page |
|
Dan Berger's chemistry pages |
|
Bluffton organic chemistry pages |
Copyright © 1998, 1999, 2000, 2003, 2007 by Daniel J. Berger. This work may be copied without limit if its use is to be for non-profit educational purposes. Such copies may be by any method, present or future. The author requests only that this statement accompany all such copies. All rights to publication for profit are retained by the author.