Lewis acids in organic chemistry
Lewis acids are often used to encourage cationic or pseudo-cationic reactions in organic chemistry. For example, silver nitrate, because of the strong affinity of Ag+ for halogens, is used to generate secondary and primary "alkyl cations" from the corresponding alkyl halides.
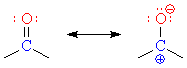 Carbonyl groups are strongly affected by Lewis acids because of their polarity. Coordination of a Lewis acid to the oxygen creates what amounts to a carbocation at the carbonyl carbon. Other oxygen- and nitrogen-containing functional groups will also bind to Lewis acids.
Carbonyl groups are strongly affected by Lewis acids because of their polarity. Coordination of a Lewis acid to the oxygen creates what amounts to a carbocation at the carbonyl carbon. Other oxygen- and nitrogen-containing functional groups will also bind to Lewis acids.
Commonly-used Lewis acids, besides silver, include titanium tetrachloride (TiCl4); zinc dichloride (ZnCl2); boron trifluoride (BF3); aluminum and alkylaluminum halides (AlX3 and RnAlX3-n); phosphorus and antimony pentafluorides (PF5 and SbF5); and tin di- and tetrachlorides (SnCl2 and SnCl4). Aluminum, antimony, silver, titanium and zinc have an affinity for halogens as well as for oxygen, while boron, tin and phosphorus are strongly attracted to oxygen. Titanium tetrachloride is used in organic synthesis as a general-purpose Lewis acid.
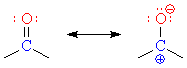 Carbonyl groups are strongly affected by Lewis acids because of their polarity. Coordination of a Lewis acid to the oxygen creates what amounts to a carbocation at the carbonyl carbon. Other oxygen- and nitrogen-containing functional groups will also bind to Lewis acids.
Carbonyl groups are strongly affected by Lewis acids because of their polarity. Coordination of a Lewis acid to the oxygen creates what amounts to a carbocation at the carbonyl carbon. Other oxygen- and nitrogen-containing functional groups will also bind to Lewis acids.